
The Impact of ICH GCP E6 Guideline R2 Revisions on Sponsors, Sites, Contract Research Organizations and Vendors | Pharmaceutical Outsourcing - The Journal of Pharmaceutical & Biopharmaceutical Contract Services
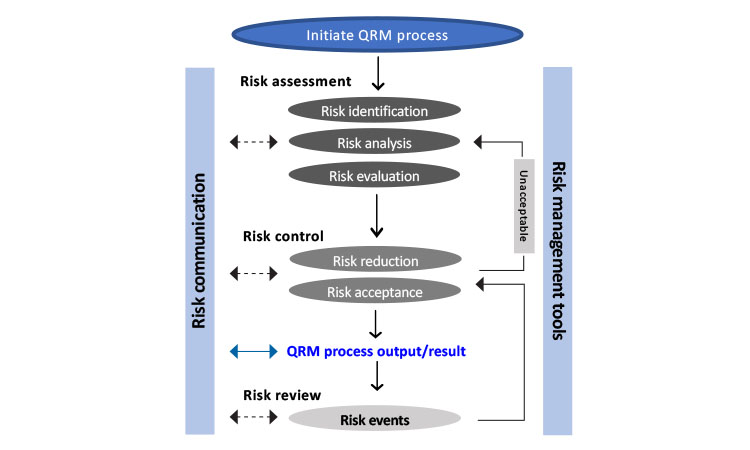
Clinical Trial Management Adaptation to ICH E6 (R2): Good Clinical Practice | Pharmaceutical Engineering

Key Differences Between Indian GCP and ICH-GCP Guidelines for Clinical Trial Conduct and Oversight | PDF




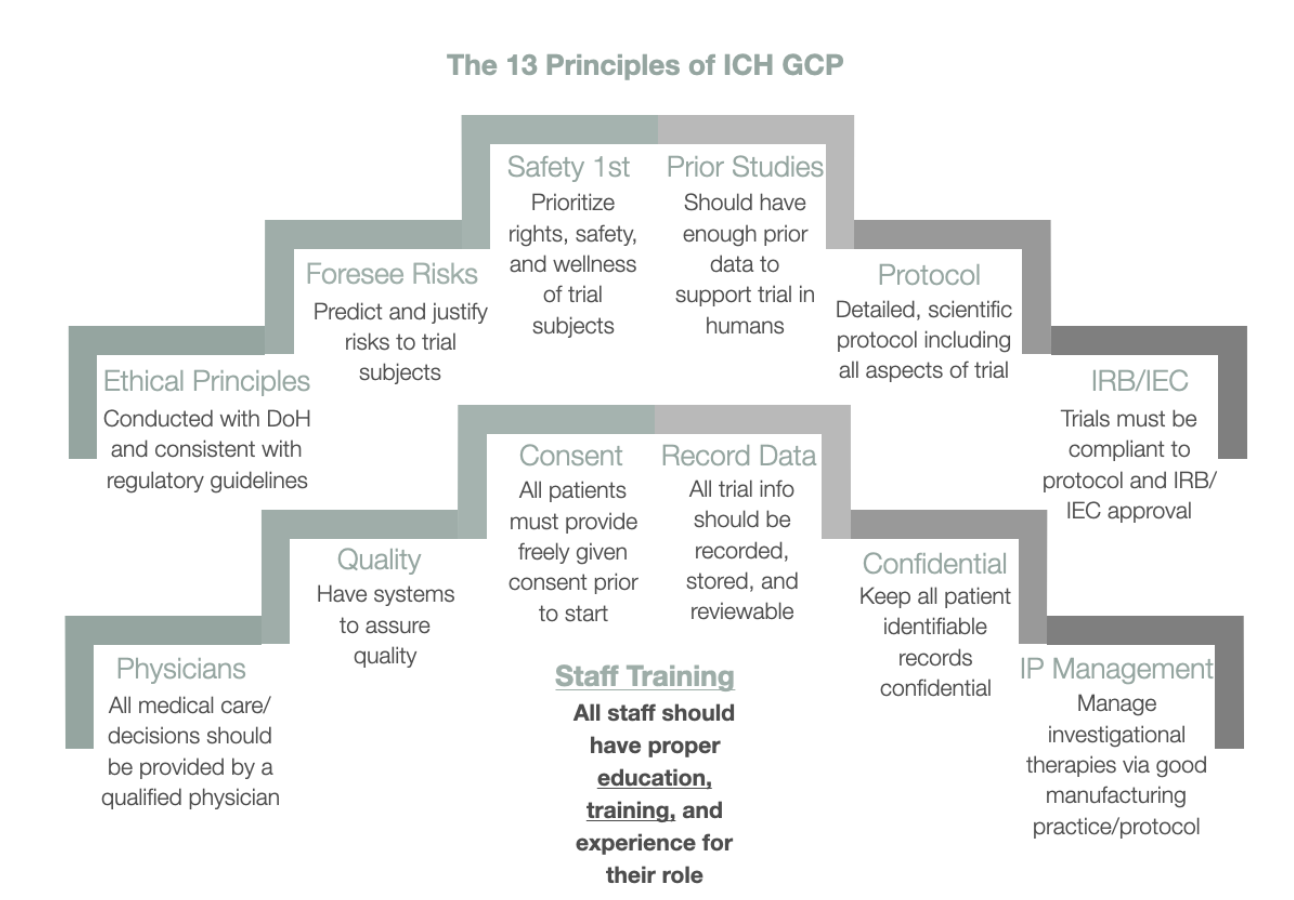

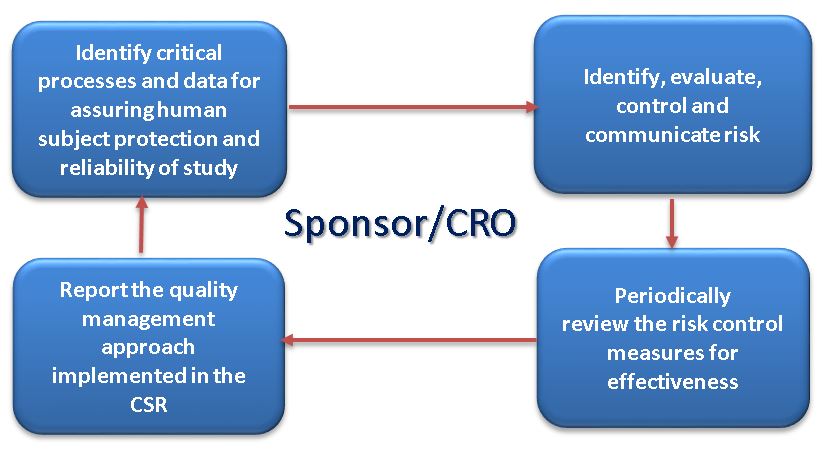



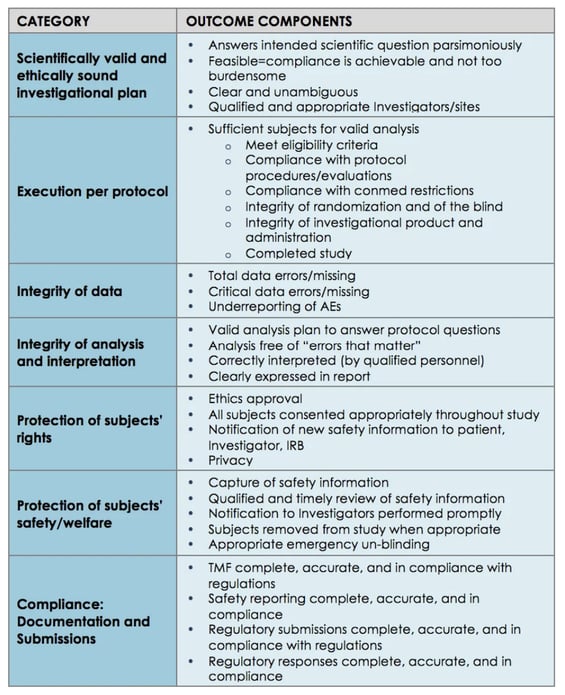






%20Guidelines.jpg?width=290&name=Statistical%20Monitoring%20in%20new%20ICH%20GCP%20E6(R2)%20Guidelines.jpg)
